

369 Innovations is a global Center of Excellence delivering FDA-ready medical device R&D and scalable contract manufacturing for healthcare leaders worldwide.
Engage Our Leadership Team
We partner with global medical device companies and growth-stage innovators
to take products from clinical insight to regulated, scalable manufacturing.
By combining FDA-ready engineering, deep manufacturing capability, and
India-based cost-efficient scale, we act as an extension of our clients’
R&D and manufacturing leadership.
369 Innovations is where global medical device strategy meets
execution excellence.
Execution-driven differentiation across the medical device lifecycle
Complete medical device development from concept definition, engineering, verification, validation, and scalable manufacturing — under one accountable partner.
Proven experience across diagnostics, patient monitoring systems, connected wearables, and hospital-grade medical devices.
A strong India-based engineering and manufacturing ecosystem delivering cost-efficient scale while meeting global quality and delivery standards.
Deep understanding of US FDA, CE, and international compliance pathways ensuring audit-ready, market-acceptable products.
End-to-End Medical Device Innovation & Manufacturing.
We provide integrated services across the medical device lifecycle — eliminating handoffs, reducing risk, and accelerating time-to-market.
We operate as a single accountable partner across the medical device lifecycle. Our services are designed for organizations that need execution certainty under regulatory, quality, and commercial constraints. From early feasibility to audit-ready manufacturing, we help our partners build devices that scale.







Innovative, validated medical devices across clinical domains

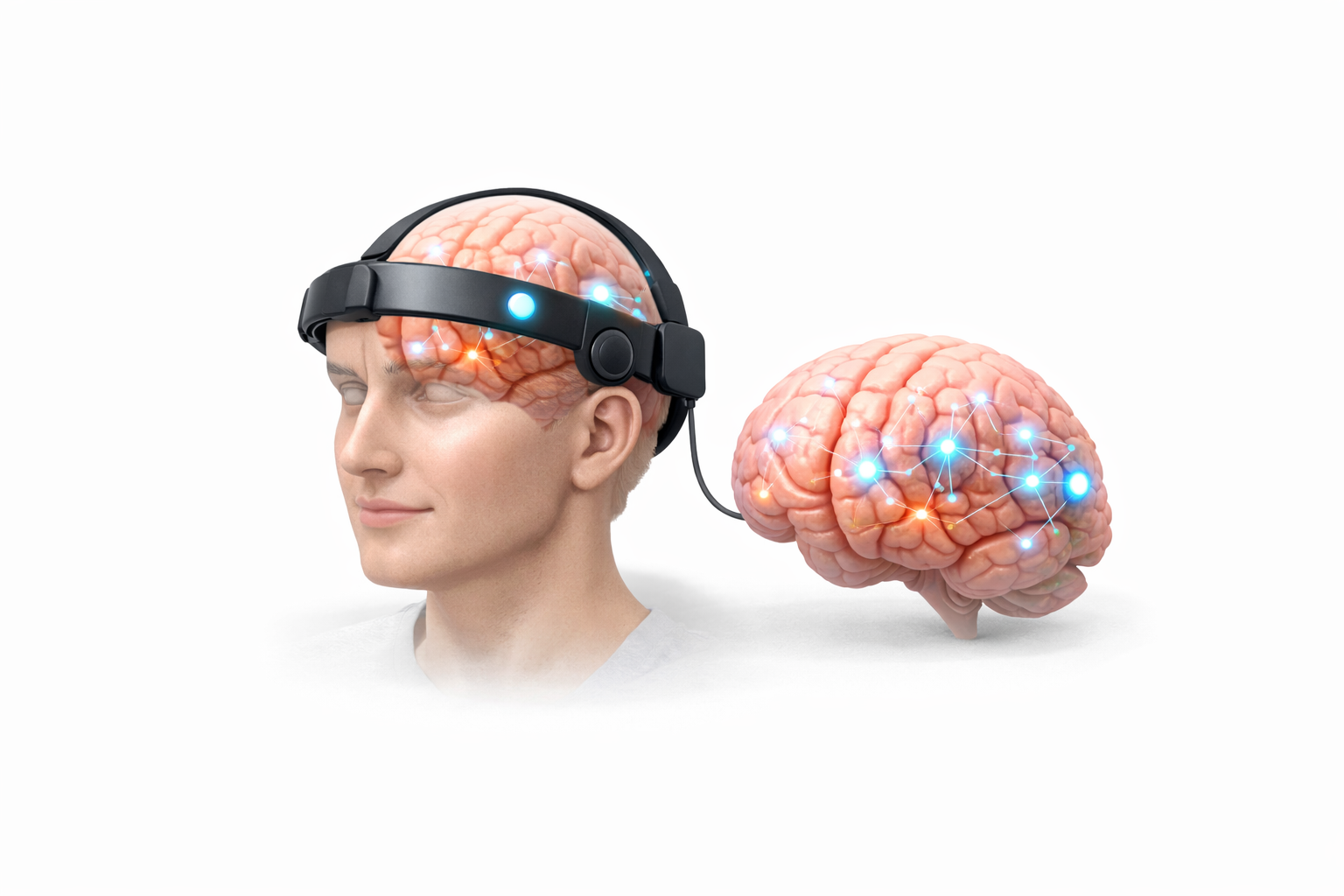
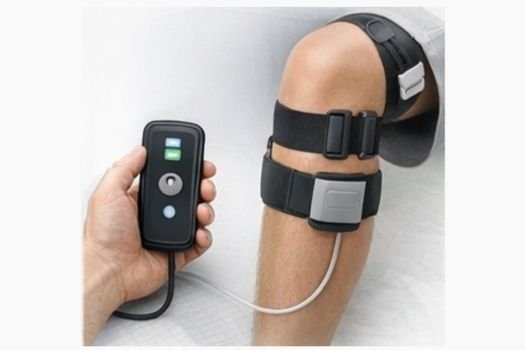

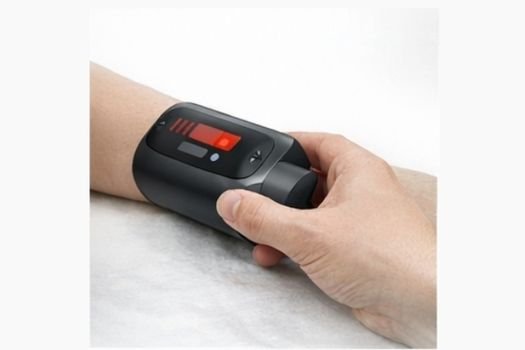
Founded by Builders. Built by Operators. Driven by Innovation.
369 Innovations was founded to address a critical gap in the medical device
ecosystem — the need for execution-focused partners who can design,
industrialize, and manufacture medical devices under real regulatory and
commercial constraints.
We are not consultants. We are builders and execution partners. Operating
as a global Center of Excellence, we work as an extension of our partners’
R&D and manufacturing leadership, delivering FDA-ready engineering and
scalable manufacturing from India to global markets.
To build one of the world’s most reliable platforms for medical device R&D and contract manufacturing — emerging from India and trusted globally.
Email: business@369innovations.com
Website: www.369innovations.com