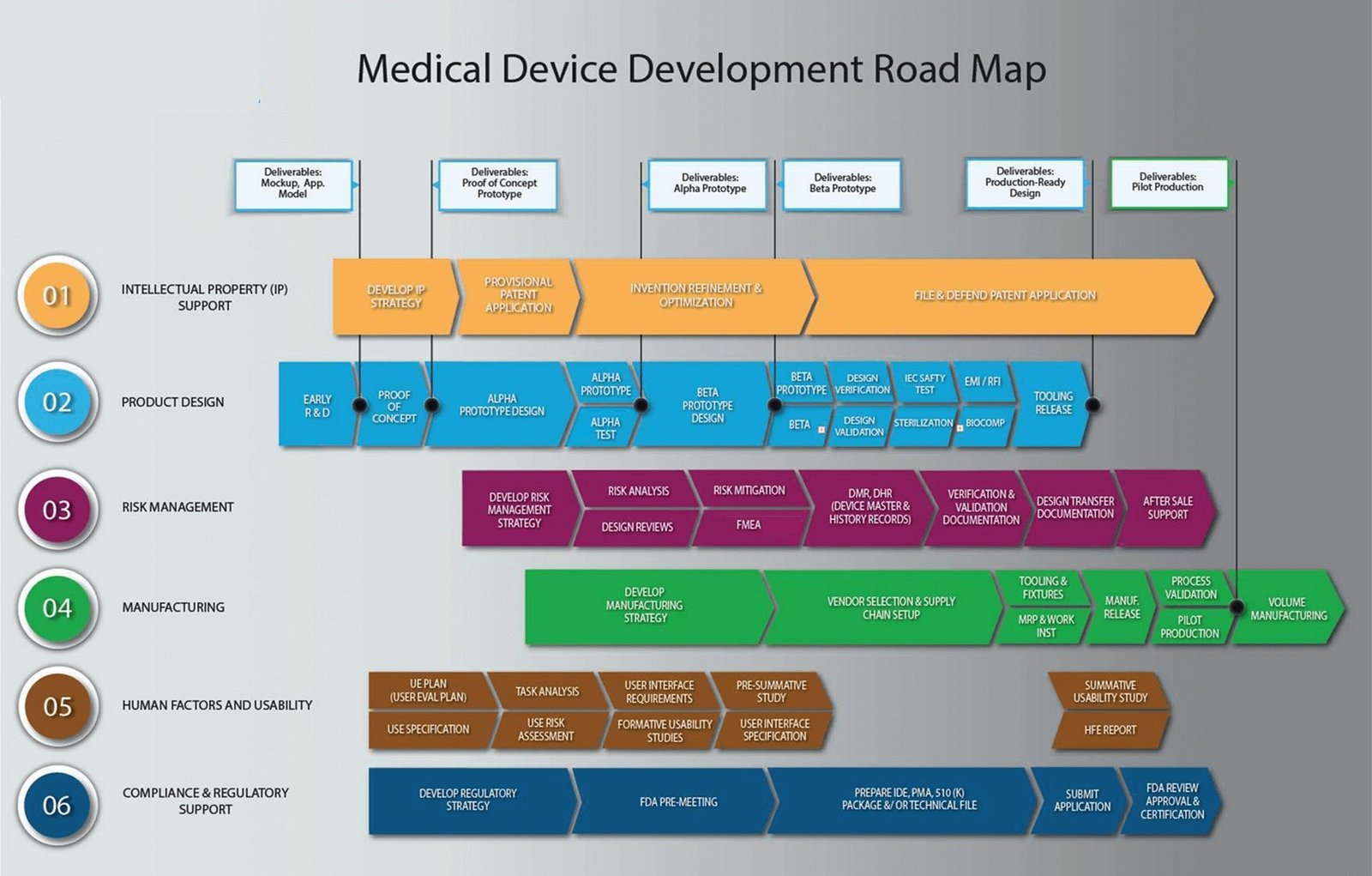
Many Dubai-based healthcare distributors rely heavily on importing finished medical devices from global brands.
But here’s the strategic question:
What if you could own your own branded device — without building a factory — in just 120 days?
With the right OEM partner, regulatory planning, and parallel execution, you can move from concept to market in approximately 120 days.
Below is a structured execution roadmap designed for GCC distributors.
Phase 1 (Days 1-30): Strategy, Product Selection & OEM Finalization
(remove device lab branding on this picture)
1 Identify the Right Product Category
Focus on:
- High-volume consumables
- Tender-driven devices
- Products with recurring demand
- Categories where margin compression is highest
Examples:
- Patient monitors
- ECG machines
- Syringe pumps
- Hospital furniture
- Disposable kits
Avoid complex Class III implantables in your first launch.
2 Select an ISO-Certified OEM Manufacturer

Ideal criteria:
- ISO 13485 certification
- CE/FDA export experience
- Prior Middle East exports
- English documentation capability
Manufacturing hubs in India like:
have strong OEM ecosystems.
Conduct:
- Virtual audit
- Quality manual review
- Sample evaluation
- NDA + supply agreement
3 Define Brand Positioning
Decide early:
- Premium vs value segment
- Target hospital segment (government vs private)
- Arabic labeling requirements
- Climate durability needs (heat, humidity)
Lock branding guidelines before regulatory filing.
Phase 2 (Days 31-60): Branding, Technical Dossier & Regulatory Preparation
4 Develop Private Label Packaging & IFU
Include:
- Arabic + English labeling
- UDI compliance (if applicable)
- Storage conditions
- Local authorized representative details
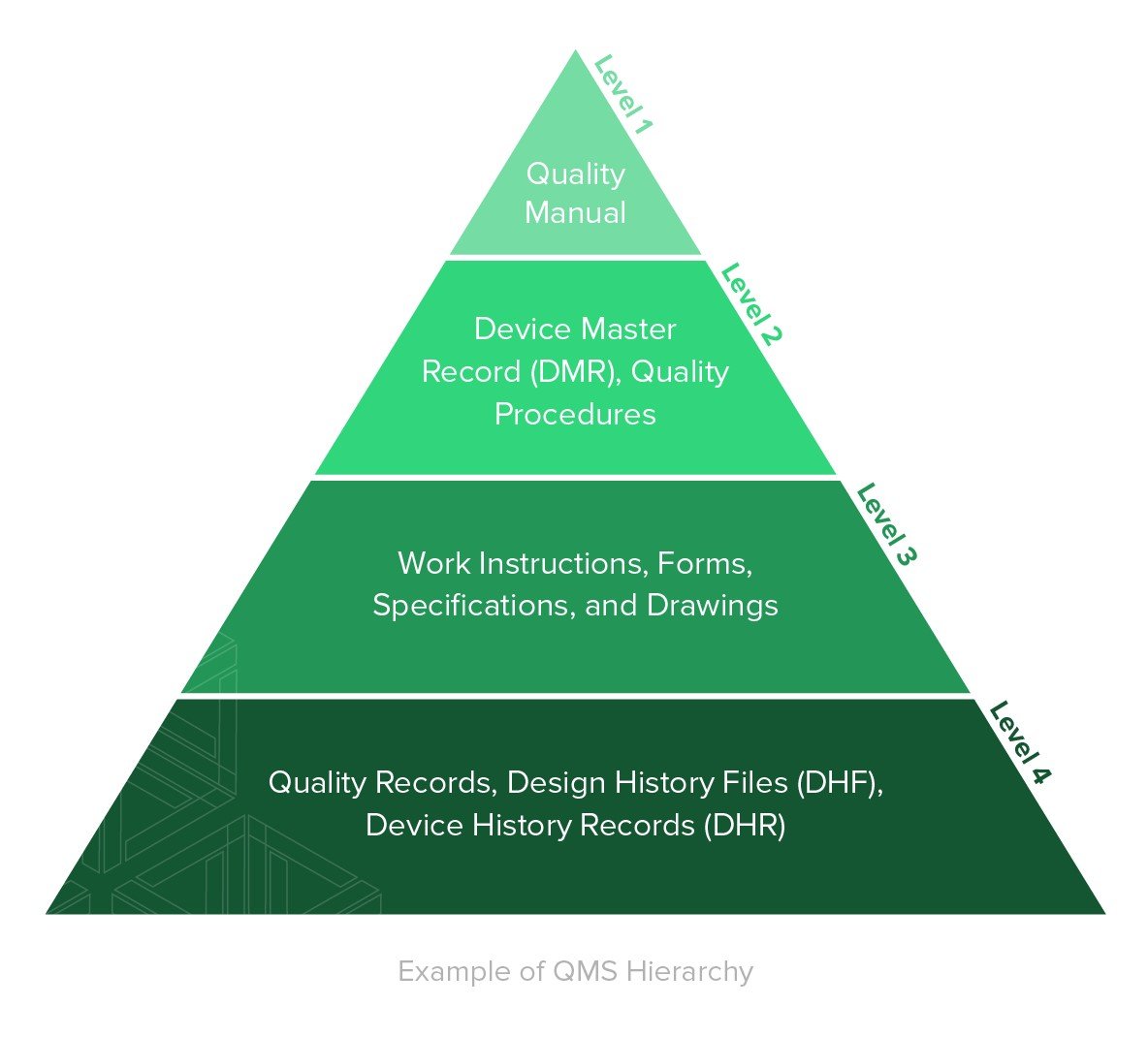
OEM typically provides:
- Device Master File
- Risk analysis
- Test reports
- CE certificate
5 Prepare Regulatory Submission (GCC-Specific)
Depending on country:
- Saudi Arabia SFDA listing
- United Arab Emirates MOHAP registration
- Qatar MOPH approval
Documents required generally:
- Free Sale Certificate
- CE certificate
- ISO 13485
- Technical file summary
- Label & artwork
Submit early approval timelines vary (30-60 days typical for lower classes).
Phase 3 (Days 61-90): Pilot Production & Market Preparation


6 Pilot Batch Manufacturing
- Produce initial MOQ
- Conduct QC inspection
- Approve final packaging
- Verify shipping cartons
Consider third-party inspection before shipment.
7 Logistics & Import Planning
- Confirm HS codes
- Verify customs clearance process
- Arrange temperature-controlled shipping (if required)
- Prepare distributor margin model
8 Sales & Tender Preparation
Parallel actions:
- Train biomedical team
- Create technical datasheets
- Develop Arabic brochures
- Prepare demo units
- Register on government tender portals
Do not wait for stock arrival to begin marketing.
Phase 4 (Days 91-120): Launch & Scale
First Shipment Arrival
- Perform incoming inspection
- Verify regulatory clearance
- Release demo stock
Market Launch Strategy
Focus on:
- Targeted hospital visits
- Government tenders
- Strategic price positioning
- Bundle pricing (equipment + consumables)
Offer:
- Extended warranty
- Local service contracts
- Competitive AMC packages
Why Private Labeling Changes the Game
Financial Snapshot (Typical Mid-Range Device)
| Component |
Estimated Allocation |
| Initial MOQ | 30- 40% capital |
| Regulatory & Documentation | 5-10% |
| Branding & Packaging | 5% |
| Logistics & Import | 10-15% |
| Sales & Demo | 10% |
Gross margin target: 25-45%
Break-even: Often achievable within first 2-3 institutional orders.
Key Risks to Manage
- Poor OEM documentation
- Underestimating regulatory timeline
- Ignoring after-sales service capacity
- Over-ordering inventory initially
- Weak quality inspection before shipment
Private label success depends on quality control + documentation discipline + service strength.
The 120-Day Acceleration Formula
- 鉁 Choose low-to-mid complexity product
- 鉁 Use CE-certified OEM
- 鉁 Run branding + regulatory in parallel
- 鉁 Start sales before stock arrival
- 鉁 Maintain tight QC
Final Thought
Launching a private label medical device is no longer a multi-year project.
With structured execution, GCC distributors can move from concept to commercial sales in 120 days with increasing margins and building long-term brand equity.







